Log in to access this content.
Free for all myUEG account holders. Your access level is set automatically based on your occupation. Medical professionals get full access to all content. If you are a non-medical user, you can only access UEG Week content from congresses you attended.
Not sure what you can access? Learn more about account types.
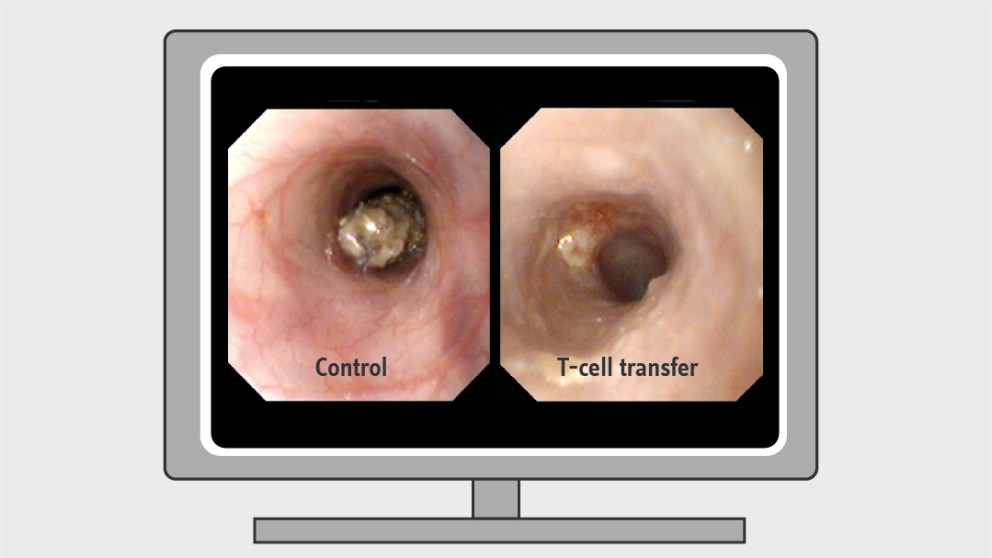
Mistakes in mouse models of IBD and how to avoid them
Topics
Citation
Published
Log in to access this content.
Free for all myUEG account holders. Your access level is set automatically based on your occupation. Medical professionals get full access to all content. If you are a non-medical user, you can only access UEG Week content from congresses you attended.
Not sure what you can access? Learn more about account types.

EAES, SAGES, and ESCP rapid guideline: bowel preparation for minimally invasive colorectal resection
Publisher
Guideline
Topics
Citation
Published
Log in to access this content.
Free for all myUEG account holders. Your access level is set automatically based on your occupation. Medical professionals get full access to all content. If you are a non-medical user, you can only access UEG Week content from congresses you attended.
Not sure what you can access? Learn more about account types.

Diagnosis and management of iatrogenic endoscopic perforations: European Society of Gastrointestinal Endoscopy (ESGE) Position Statement – Update 2020
Publisher
Guideline
Topics
Citation
Published
Log in to access this content.
Free for all myUEG account holders. Your access level is set automatically based on your occupation. Medical professionals get full access to all content. If you are a non-medical user, you can only access UEG Week content from congresses you attended.
Not sure what you can access? Learn more about account types.

UEG and EAES rapid guideline: Systematic review, meta-analysis, GRADE assessment and evidence-informed European recommendations on TaTME for rectal cancer
Publisher
Guideline
Topics
Citation
Published

Log in to access this content.
Free for all myUEG account holders. Your access level is set automatically based on your occupation. Medical professionals get full access to all content. If you are a non-medical user, you can only access UEG Week content from congresses you attended.
Not sure what you can access? Learn more about account types.
Microbiome: Implications for clinicians - gut-liver and gut-pancreas axis
1 Universitätsklinikum Göttingen, Hamburg, Germany
2 Fondazione Policlinico Universitario 'A.Gemelli' - IRCCS, MONTEROTONDO - ROME, Italy
Event
Citation
Published
Log in to access this content.
Free for all myUEG account holders. Your access level is set automatically based on your occupation. Medical professionals get full access to all content. If you are a non-medical user, you can only access UEG Week content from congresses you attended.
Not sure what you can access? Learn more about account types.

Endoscopic management of superficial nonampullary duodenal tumors: European Society of Gastrointestinal Endoscopy (ESGE) Guideline
Publisher
Guideline
Topics
Citation
Published
Log in to access this content.
Free for all myUEG account holders. Your access level is set automatically based on your occupation. Medical professionals get full access to all content. If you are a non-medical user, you can only access UEG Week content from congresses you attended.
Not sure what you can access? Learn more about account types.

European Society of Coloproctology guideline on training in robotic colorectal surgery (2024)
Publisher
Guideline
Topics