Log in to access this content.
Free for all myUEG account holders. Your access level is set automatically based on your occupation. Medical professionals get full access to all content. If you are a non-medical user, you can only access UEG Week content from congresses you attended.
Not sure what you can access? Learn more about account types.
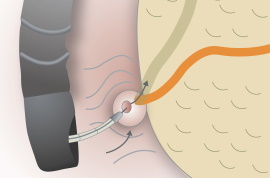
Endoscopic retrograde cholangiopancreatography (ERCP)
Event
Topics
Accreditation status
Duration
Published
Log in to access this content.
Free for all myUEG account holders. Your access level is set automatically based on your occupation. Medical professionals get full access to all content. If you are a non-medical user, you can only access UEG Week content from congresses you attended.
Not sure what you can access? Learn more about account types.
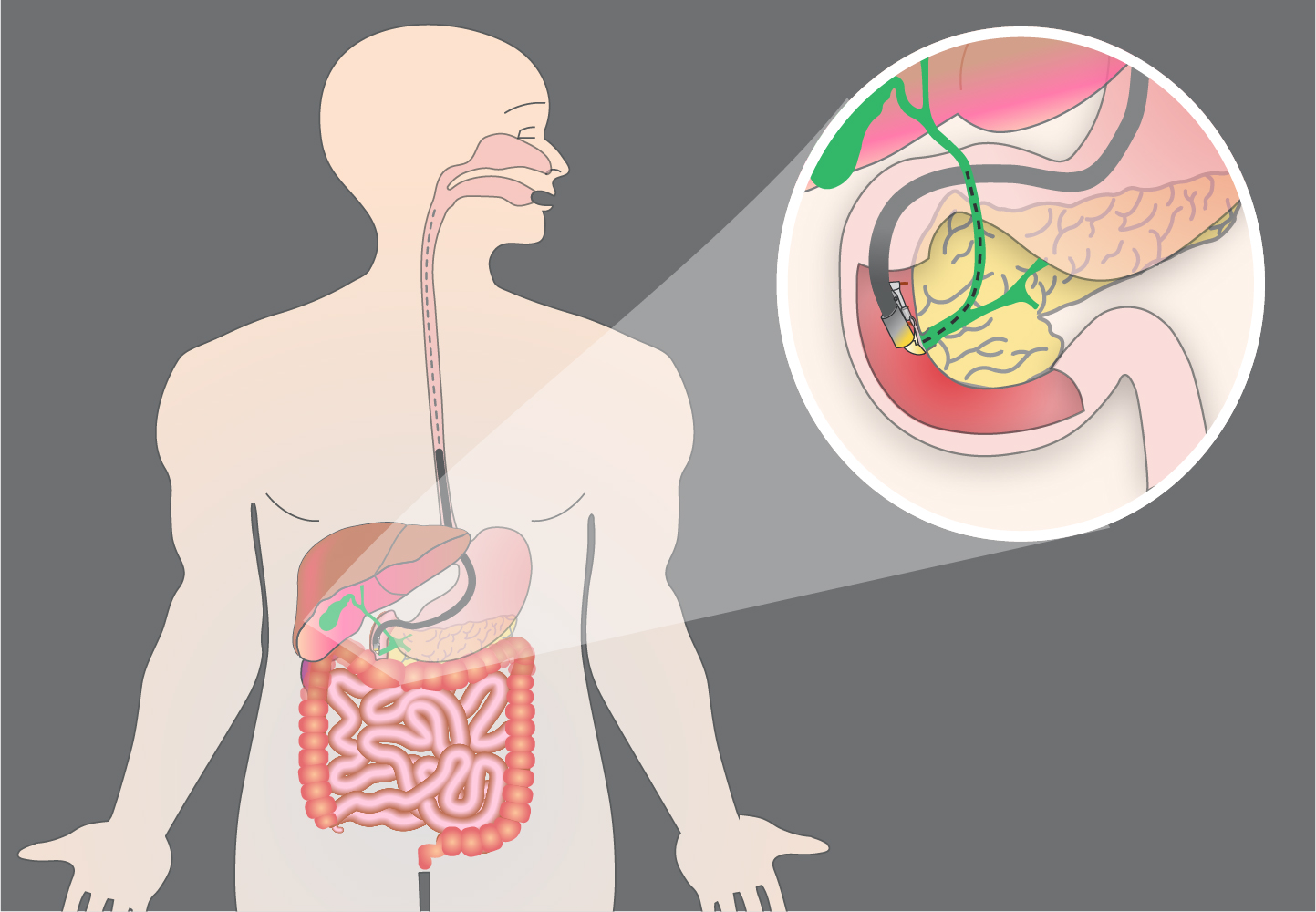
Advanced ERCP - Part 1
Event
Topics
Accreditation status
Duration
Published
Log in to access this content.
Free for all myUEG account holders. Your access level is set automatically based on your occupation. Medical professionals get full access to all content. If you are a non-medical user, you can only access UEG Week content from congresses you attended.
Not sure what you can access? Learn more about account types.

WHAT MOTIVATES MEDICAL INTERNS TO PURSUE IN GASTROENTEROLOGY? A MULTICENTRIC CROSS-SECTIONAL STUDY
Amine Achemlal 1, sanaa berrag 1, Chaimae Haddad Hachimi 1, Salma Azammam 1, Meriem Amine 1, Sakina Oualaalou 1, salma ouahid 1, Rachid Laroussi 1, Nejjari Fouad 1, Tarik ADIOUI 1, Mouna Tamzaourte 1
1 Mohamed V Military Training Hospital, Rabat, Morocco
Conference
Submission format
Session
Citation
Published
Log in to access this content.
Free for all myUEG account holders. Your access level is set automatically based on your occupation. Medical professionals get full access to all content. If you are a non-medical user, you can only access UEG Week content from congresses you attended.
Not sure what you can access? Learn more about account types.

EUROPEAN AND UNITED STATES PATIENT PERSPECTIVES ON THE IMPACT OF MODERATE TO SEVERE ULCERATIVE COLITIS ON SEXUAL ACTIVITY: COMMUNICATING NEEDS AND FEATURES OF IBD EXPERIENCES (CONFIDE) SURVEY
Simon Travis 1, Alison Potts Bleakman 2, Marla C. Dubinsky 3, David T. Rubin 4, Remo Panaccione 5, Toshifumi Hibi 6, Cem Kayhan 2, Theresa Hunter Gibble 2, Christophe Sapin 2, Eoin J. Flynn 2, Christian Atkinson 7, Stefan Schreiber 8
1 University of Oxford, Oxford, United Kingdom
2 Eli Lilly and Company, Indianapolis, United States
3 Icahn School of Medicine, New York City, United States
4 University of Chicago Medicine Inflammatory Bowel Disease Center, Chicago, United States
5 University of Calgary, Calgary, Canada
6 Kitasato University, Tokyo, Japan
7 Adelphi Real World, Bollington, United Kingdom
8 University Hospital Schleswig-Holstein, Kiel, Germany
Conference
Topics
Submission format
Session
Citation
Published
Log in to access this content.
Free for all myUEG account holders. Your access level is set automatically based on your occupation. Medical professionals get full access to all content. If you are a non-medical user, you can only access UEG Week content from congresses you attended.
Not sure what you can access? Learn more about account types.

EX VIVO EFFECTS OF INFLIXIMAB ON THE LONG NON-CODING RNAS EXPRESSION LEVELS IN CROHN´S DISEASE
Montse Baldan Martin 1, Cristina Rubín de Célix 1, Macarena Orejudo del Río 1, Lorena Ortega Moreno 2, Samuel Fernández-Tomé 3, Irene Soleto 1, Cristina Ramírez 1, Ricardo Arroyo 4, Paloma Fernández 4, Cecilio Santander Vaquero 1, Jose Andres Moreno-Monteagudo 1, María José Casanova 1, Fernando Casals Seoane 1, Sergio Casabona-Francés 1, Irene Becerro Gonzalez 1, Urko M. Marigorta 5, Ana M. Aransay 6, David Bernardo 7, María Chaparro 1, Javier Gisbert 1
1 Hospital Universitario de La Princesa, Instituto de Investigación Sanitaria Princesa (IIS-Princesa), Universidad Autónoma de Madrid (UAM), and Centro de Investigación Biomédica en Red de Enfermedades Hepáticas y Digestivas (CIBERehd), Madrid, Spain
2 Área de Farmacología y Nutrición y Bromatología, Universidad Rey Juan Carlos, Madrid, Spain
3 Hospital Universitario de La Princesa, Instituto de Investigación Sanitaria Princesa (IIS-Princesa), Universidad Autónoma de Madrid (UAM), and Centro de Investigación Biomédica en Red de Enfermedades Hepáticas y Digestivas (CIBERehd), Madrid, Spain|||Facu
4 Instituto de Medicina Molecular Aplicada Nemesio Díez (IMMA-ND), Facultad de Medicina. Universidad San Pablo CEU, Madrid, Spain
5 Integrative Genomics Lab, CIC bioGUNE-BRTA and IKERBASQUE, Basque Foundation for Science, Bilbao, Spain
6 Genome Analysis Platform, CIC bioGUNE-BRTA and CIBERehd, Bilbao, Spain
7 Mucosal Immunology Lab, Unidad de Excelencia Instituto de Biomedicina y Genética Molecular (IGBM, Universidad de Valladolid-CSIC) and Centro de Investigación Biomédica en Red de Enfermedades Infecciosas (CIBERINFEC), Valladolid, Spain
Conference
Topics
Submission format
Session
Citation
Published
Log in to access this content.
Free for all myUEG account holders. Your access level is set automatically based on your occupation. Medical professionals get full access to all content. If you are a non-medical user, you can only access UEG Week content from congresses you attended.
Not sure what you can access? Learn more about account types.

UTILITY OF RADIOLOGICAL FOLLOW UP OF MAIN-DUCT INTRADUCTAL PAPILLARY MUCINOUS NEOPLASMS AND MIXED-TYPE INTRADUCTAL PAPILLARY MUCINOUS NEOPLASMS
Roie Tzadok 1, Rivka Kessner 1, Nir Bar 1, Hila Yashar 1, Sapir Lazar 1, Yuval Katz 1, Roie Tzadok 1, Arthur Chernomorets 1, Oren Shibolet 1, Dana Ben-Ami Shor 1
1 Tel Aviv Sourasky Medical Center, Tel Aviv, Israel
Conference
Topics
Submission format
Session
Citation
Published
Log in to access this content.
Free for all myUEG account holders. Your access level is set automatically based on your occupation. Medical professionals get full access to all content. If you are a non-medical user, you can only access UEG Week content from congresses you attended.
Not sure what you can access? Learn more about account types.

Mistakes in endoscopic retrograde cholangiopancreatography and how to avoid them
Topics